Bentley is a German-based medtech company with a well-positioned and innovative portfolio of covered and uncovered stents and catheters for endovascular therapies.
Supplying the markets with high-quality products and the largest portfolio of covered stents, Bentley focuses today on peripheral arterial diseases (PAD) and on future-oriented projects on the treatment of aortic aneurysms. Bentley has a strong market positioning in the assessed segments, balloon-expandable covered stents and peripheral CTO (chronic total occlusion) devices.
The total market for balloon-expandable covered stents and CTO devices in 2023 is estimated at EUR 940 million. The markets are expected to grow from the increasing diagnosis and adoption rates in the Americas and EMEA (Europe, Middle East and Africa) and the demographic shifts and increasing disease prevalence in APAC (Asia Pacific). With future indication expansions and strategic acquisitions additionally benefiting the existing portfolio, Bentley will keep its strong market position and grow further, focusing on product quality, innovation capabilities, and strong customer relationships.
Best Quality for Top-Level Physicians
Our Bentley products have unique features complemented by a large variety of stent sizes that makes them the first choice for physicians. As a result, our high-quality products enjoy the highest reputation for minimally invasive procedures to treat vascular diseases.










BeGraft aortic
The BeGraft aortic stent graft system provides low foreshortening, flexibility and radial force where it is needed, e.g., for restoring and improving the patency of the iliac arteries. It is approved for treating coarctation of the descending aorta and is a lifesaving solution in the field of pediatric cardiology.

BeGraft coronary
This reliable product combines a highly flexible stent graft with the lowest-profile balloon catheter to assist in emergency situations.

BeGraft peripheral
This peripheral stent graft system excels with a high radial force while maintaining flexibility and a low profile for challenging arterial anatomies.

BeGraft+ peripheral
This peripheral stent graft uniquely combines an extraordinary radial force through our unique sandwich design with remarkable flexibility.

BeSmooth peripheral
The BeSmooth peripheral stent system stands out with its low profile that ensures 6F compatibility for all sizes. Its high flexibility is designed to provide good vessel adaption.

BeYond venous
The BeYond venous stent is designed to respect the natural venous anatomy to ensure the optimal balance between radial force, flexibility and deployment behavior.
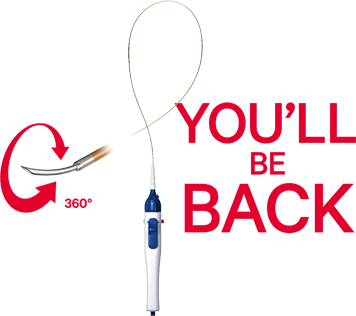
BeBack
The BeBack crossing catheter is a low-profile support catheter that has a crossing and targeted re-entry capability with a length-adjustable, 360° rotational Nitinol needle. The special radiopaque marker indicates in which direction the curved needle protrudes from the tip of the catheter.